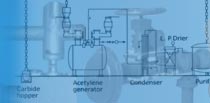
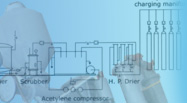
| Process
Description
of KVK Oxygen/Nitrogen gas plant |
Atmospheric
air mainly consists of oxygen and nitrogen gases along with
small quantities of water vapour, carbon dioxide, argon, helium,
etc. Oxygen and Nitrogen from the air are separated due to difference
in boiling points by distillation through a fractional column.
Atmospheric air is sucked in by a multi stage compressor through
a filter and compressed to the design pressure. The compressed
air is then passed through inter-coolers, industrial refrigerator,
moisture separators, and then to the molecular sieve battery
for removal of Carbon dioxide, hydrocarbons and moisture from
the process air. This pure air then passes through the 1st heat
exchanger, where it is cooled by the out going nitrogen and
oxygen. Part of this cooled air then passed through an Expansion
Engine and the other part through the 2nd heat exchanger. Both
the Expansion Engine and 2nd heat exchanger help in further
cooling down the air, which is finally released to the bottom
of the column through an expansion valve. The air becomes liquid
at this stage. |
|
The
column consists of two parts. The lower column and upper column.
In between the two, lower and upper columns there is a condenser,
which acts as a reflux for the lower column and as a re-boiler
for the upper column. The liquid air at the bottom of lower
column separates through the trays of the column to give crude
oxygen at the bottom and approximately 90% pure nitrogen at
the top. Crude oxygen termed as rich liquid is then expanded
through an expansion valve from the lower column to the middle
of the upper column. Crude nitrogen termed as poor liquid
is expanded through another expansion valve from the top lower
column to the top of the upper column. Due to difference in
the boiling points, the pure nitrogen boils over and accumulates
at the top of the upper column and oxygen (which has a higher
boiling point then nitrogen) accumulates at the bottom of
the upper column.
Both nitrogen and oxygen are removed through separate paths
in heat exchangers, for cooling the incoming air. Oxygen is
compressed to a prescribed settled pressure by a liquid pump
and is directly filled into cylinders. Nitrogen is however
available at a pressure of a approximately 0.5kg/cm2 and the
same can be compressed into cylinders with help of an independent
high- pressure compressor.
Normally the nitrogen is of 97.5 to 98.5% (Approx) purity
only and is let out into the atmosphere as waste product.
However, when nitrogen is to filled, into the cylinders for
commercial use, the plant is controlled by a change in the
valve setting to make the mixed air chamber operative to achieve
the required nitrogen purity. (This is a specialty of our
design).
|
| Top |
|
|